|
Chemical bonding is the backbone that defines a molecule complex’s internal structure and nature and characteristics (both physical and chemical). Molecules are generated through the formation of certain bonds between atoms that are based on their strength. Homogeneous and heterogeneous molecules are formed when two or more atoms react and combine. XeF 2 Molecular Geometry: Chemical Adhesion It is left with three lone pairs placed in the equatorial plane and two fluorines arranged perpendicularly to the lone pairs in the axial plane after sharing 1–1 electron with fluorine. 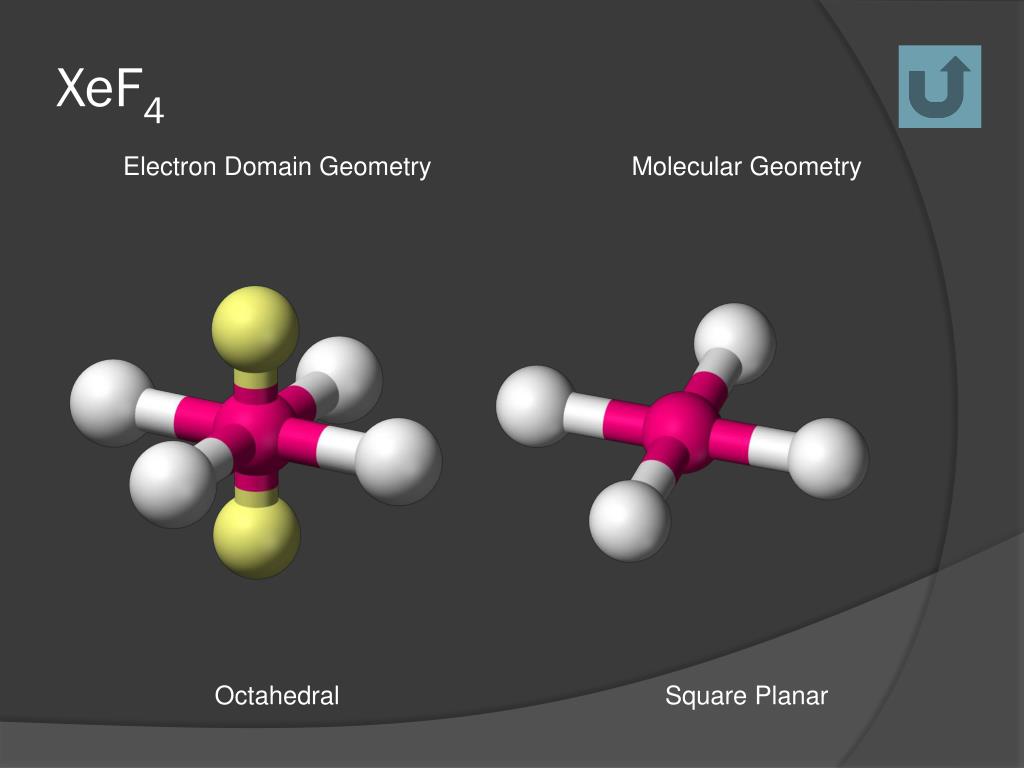
Due to the 180-degree bond angles, the structure must be linear. Although the lone pairs are at equatorial positions (bonds perpendicular to the axis), the molecule is a trigonal bipyramid. This indicates that both fluorines must be bound to the Xe molecule, resulting in three unshared pairs and two bonded pairs on the Xe molecule. Molecule Xe has eight valence electrons, while fluorine has seven, totalling 22 valence electrons. 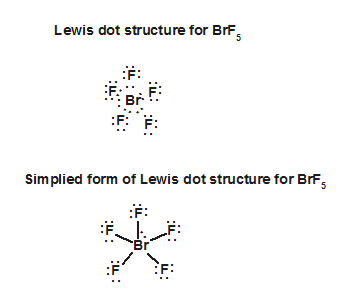
XeF 2 molecular geometry is an important and interesting topic. XeF 2 is generated when a 2:1 mixture of xenon and fluorine is heated at 400 ℃ in a sealed nickel tank. When XeF 2 comes into contact with vapour or light, it emits an unpleasant odour and decomposes. Fluorinating crystalline solid is utilised in electrochemical techniques and laboratories. XeF 2 is the most stable of the three chemicals. Other Xenon compounds include XeF 4 (Xenon Tetrafluoride) and XeF 6 ( Xenon Hexafluoride). It’s a fluorinating and oxidising agent with a lot of punch. 
The chemical compound Xenon Difluoride is abbreviated as XeF 2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
